Chemistry Latex
Test Section Summary
0 of 136 Questions completed
Questions:
Information
You have already completed the test section before. Hence you can not start it again.
Test Section is loading…
You must sign in or sign up to start the test section.
You must first complete the following:
Results
Results
0 of 136 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Acids and Bases 0%
- Chemical Bonds 0%
- Chemical Equations 0%
- Chemical Solutions 0%
- Properties of Matter 0%
- Reaction Rate 0%
- Scientific Notation 0%
- States of Matter 0%
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36
- 37
- 38
- 39
- 40
- 41
- 42
- 43
- 44
- 45
- 46
- 47
- 48
- 49
- 50
- 51
- 52
- 53
- 54
- 55
- 56
- 57
- 58
- 59
- 60
- 61
- 62
- 63
- 64
- 65
- 66
- 67
- 68
- 69
- 70
- 71
- 72
- 73
- 74
- 75
- 76
- 77
- 78
- 79
- 80
- 81
- 82
- 83
- 84
- 85
- 86
- 87
- 88
- 89
- 90
- 91
- 92
- 93
- 94
- 95
- 96
- 97
- 98
- 99
- 100
- 101
- 102
- 103
- 104
- 105
- 106
- 107
- 108
- 109
- 110
- 111
- 112
- 113
- 114
- 115
- 116
- 117
- 118
- 119
- 120
- 121
- 122
- 123
- 124
- 125
- 126
- 127
- 128
- 129
- 130
- 131
- 132
- 133
- 134
- 135
- 136
- Current
- Review
- Answered
- Correct
- Incorrect
-
Question 1 of 136
1. Question
What is the conjugate acid in the following equation?
CH₃COOH + H₂ O ⇌ H₃O^+ +CH₃COO^-
CorrectIncorrect -
Question 2 of 136
2. Question
Which would turn litmus paper blue?
CorrectIncorrect -
Question 3 of 136
3. Question
What happens to a Brnsted-Lowry base in an aqueous solution?
CorrectIncorrect -
Question 4 of 136
4. Question
Which product completes the following acid-base reaction?
H₂ SO₄+2KOH → ?
CorrectIncorrect -
Question 5 of 136
5. Question
What is the conjugate base of sulfuric acid?
CorrectIncorrect -
Question 6 of 136
6. Question
What is formed when nitric acid (HNO₃) reacts with barium hydroxide (Ba(OH)₂)?
CorrectIncorrect -
Question 7 of 136
7. Question
Which of the following is an acid?
CorrectIncorrect -
Question 8 of 136
8. Question
Which ions from a base react with hydrogen ions in a neutralization reaction?
CorrectIncorrect -
Question 9 of 136
9. Question
Using the following pH scale, which substance would be slippery to the touch when dissolved in water?
 CorrectIncorrect
CorrectIncorrect -
Question 10 of 136
10. Question
What product is formed when acetic acid (CH₃COOH) reacts with water?
CorrectIncorrect -
Question 11 of 136
11. Question
What conjugate base is produced when the Brnsted-Lowry acid hydrogen phosphate \((HPO_4^{2-})\) reacts with a hydroxide ion?
CorrectIncorrect -
Question 12 of 136
12. Question
Which is a characteristic of a basic solution?
CorrectIncorrect -
Question 13 of 136
13. Question
What is the conjugate base in the following reaction?
HNO₂+H₂O ↔ ?
CorrectIncorrect -
Question 14 of 136
14. Question
Using the following image, what substance is most likely a weak acid?
 CorrectIncorrect
CorrectIncorrect -
Question 15 of 136
15. Question
When HNO₃ reacts with NaOH, which product is formed?
CorrectIncorrect -
Question 16 of 136
16. Question
What is the conjugate acid of S²⁻?
CorrectIncorrect -
Question 17 of 136
17. Question
Which partially dissociates into hydroxide ions in an aqueous solution?
CorrectIncorrect -
Question 18 of 136
18. Question
An acid that dissociates into hydrogen ions is defined as a(n)
CorrectIncorrect -
Question 19 of 136
19. Question
Which of the following is a strong acid?
CorrectIncorrect -
Question 20 of 136
20. Question
Which of the following would completely ionize in solution?
CorrectIncorrect -
Question 21 of 136
21. Question
In which compound(s) does a chlorine atom share two electrons with another atom?
CorrectIncorrect -
Question 22 of 136
22. Question
Octane in gasoline has the formula C₈H₁₈.
What type of compound is it, and why?
CorrectIncorrect -
Question 23 of 136
23. Question
In the molecule of chlorine (Cl₂) shown below, how is each chlorine atom stable?
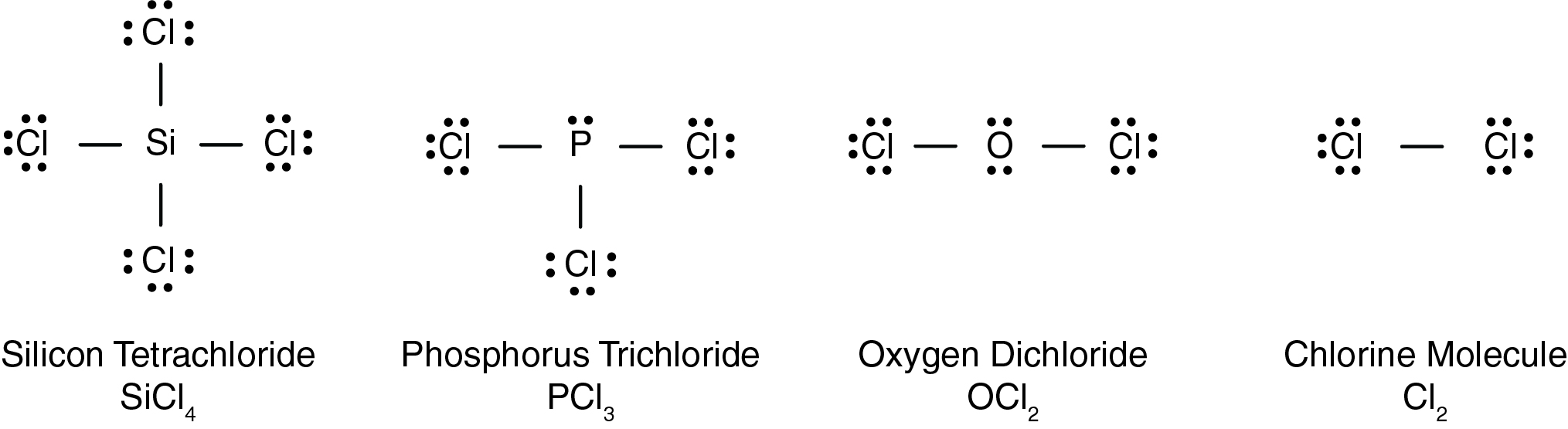 CorrectIncorrect
CorrectIncorrect -
Question 24 of 136
24. Question
In the compound silicon tetrachloride (SiCl₄) shown below, how is silicon stable?
 CorrectIncorrect
CorrectIncorrect -
Question 25 of 136
25. Question
In the Lewis structures below, which element is stable with four shared and four lone-pair electrons?
 CorrectIncorrect
CorrectIncorrect -
Question 26 of 136
26. Question
Consider the carbon-carbon bonds that exist in the molecules shown below.
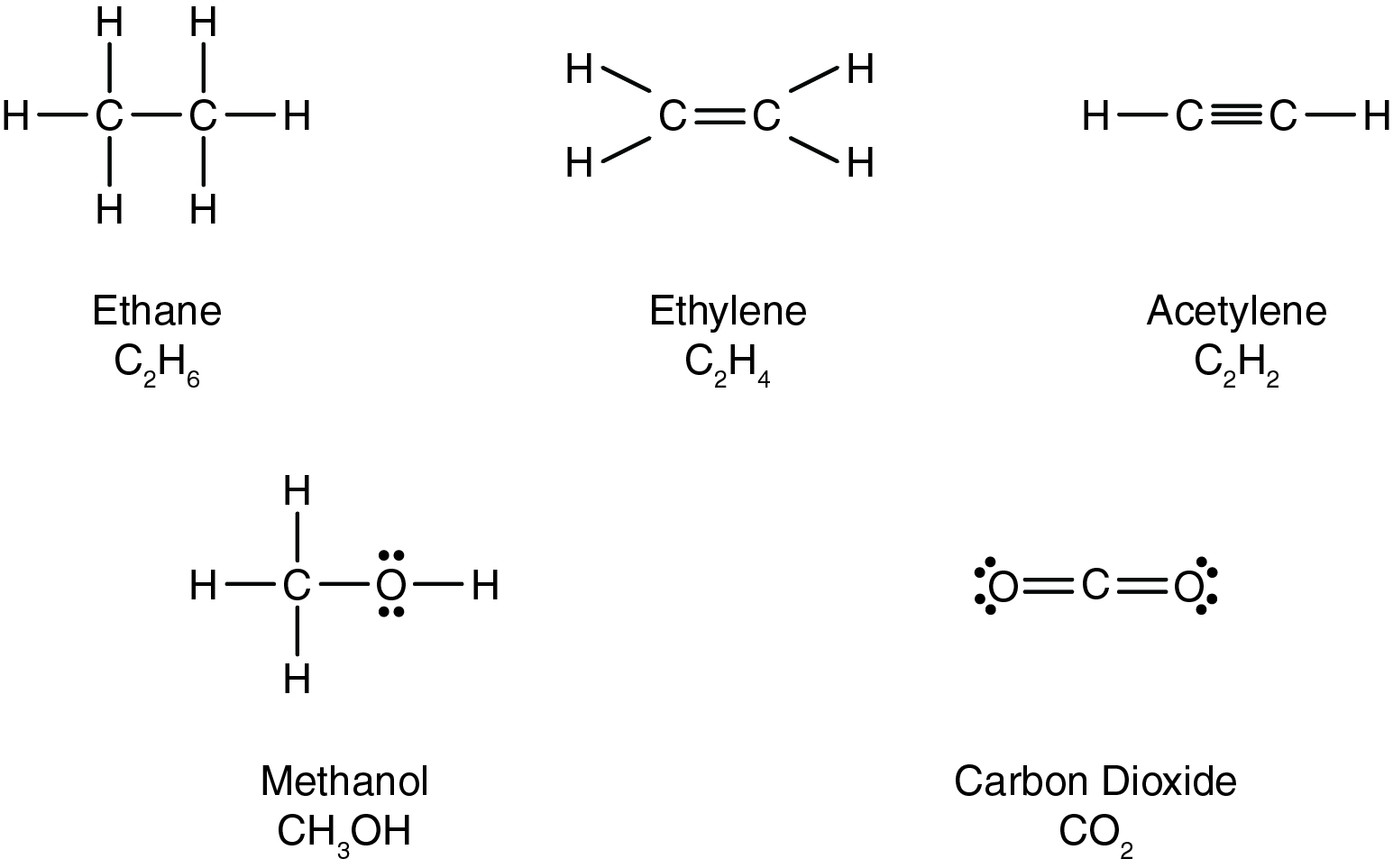
Which of the compounds below contains the strongest carbon-carbon bond?
CorrectIncorrect -
Question 27 of 136
27. Question
According to the Lewis structure below, what is the formula for methanol?
 CorrectIncorrect
CorrectIncorrect -
Question 28 of 136
28. Question
The compound silver sulfide has the formula Ag₂S.
Which of the following best describes the meaning of the subscript 2 in the formula?
CorrectIncorrect -
Question 29 of 136
29. Question
In the compound sodium bromide (NaBr), electrons are _____.
In the compound carbon tetrabromide (CBr₄), electrons are _____.
CorrectIncorrect -
Question 30 of 136
30. Question
What is the net charge of the ionic compound barium iodide (BaI₂)?
CorrectIncorrect -
Question 31 of 136
31. Question
In the Lewis structure of a fluorine molecule shown below, how are the eight valence electrons of each fluorine atom arranged?
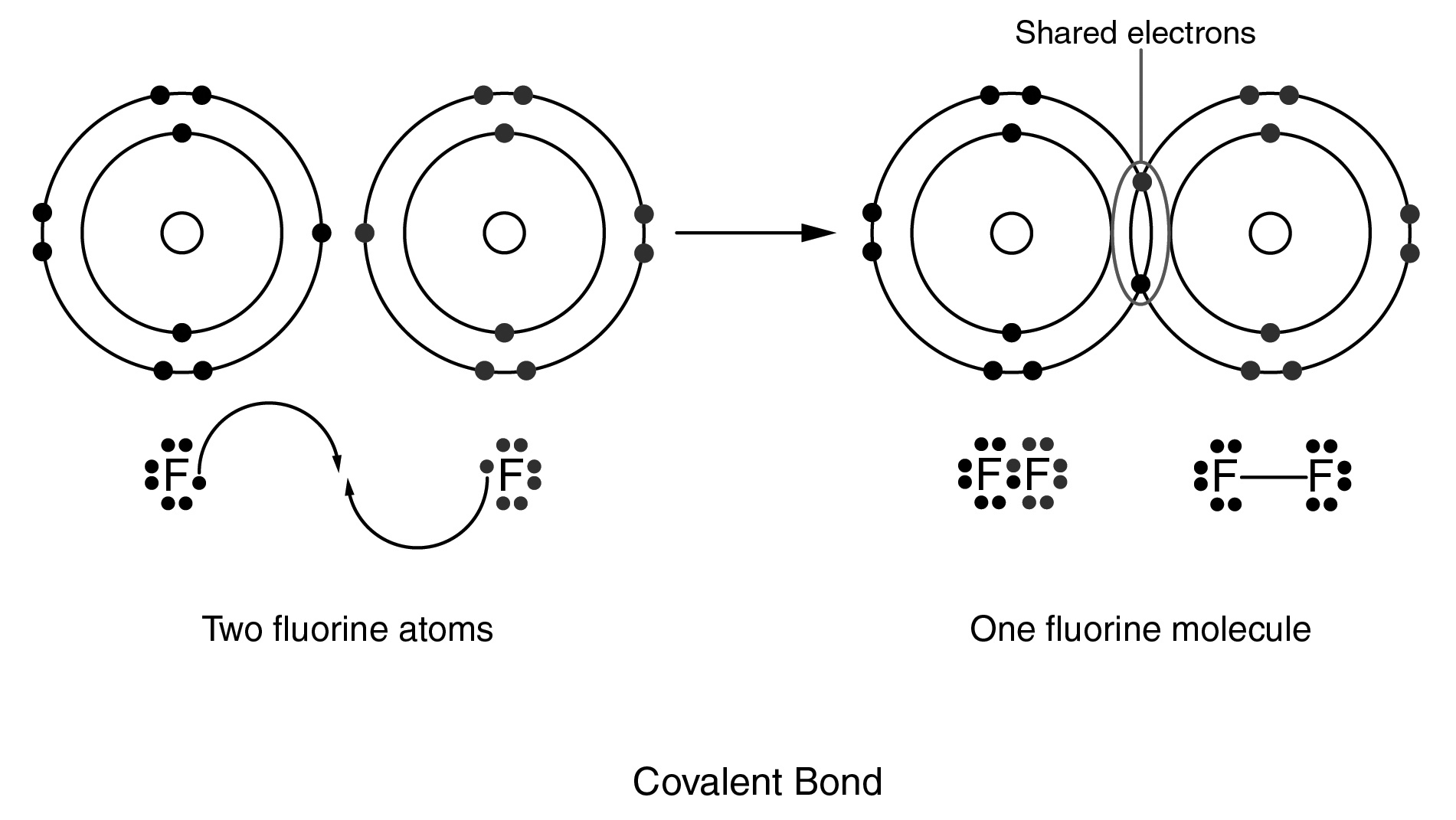 CorrectIncorrect
CorrectIncorrect -
Question 32 of 136
32. Question
Out of the elements present in the Lewis structures below, which one has the greatest number of shared electrons? Consider only single atoms of each element.
 CorrectIncorrect
CorrectIncorrect -
Question 33 of 136
33. Question
What is the formula for the compound formed between calcium and oxygen?
CorrectIncorrect -
Question 34 of 136
34. Question
In the Lewis structure for carbon dioxide shown below, how many electrons is the carbon atom sharing with one oxygen atom?
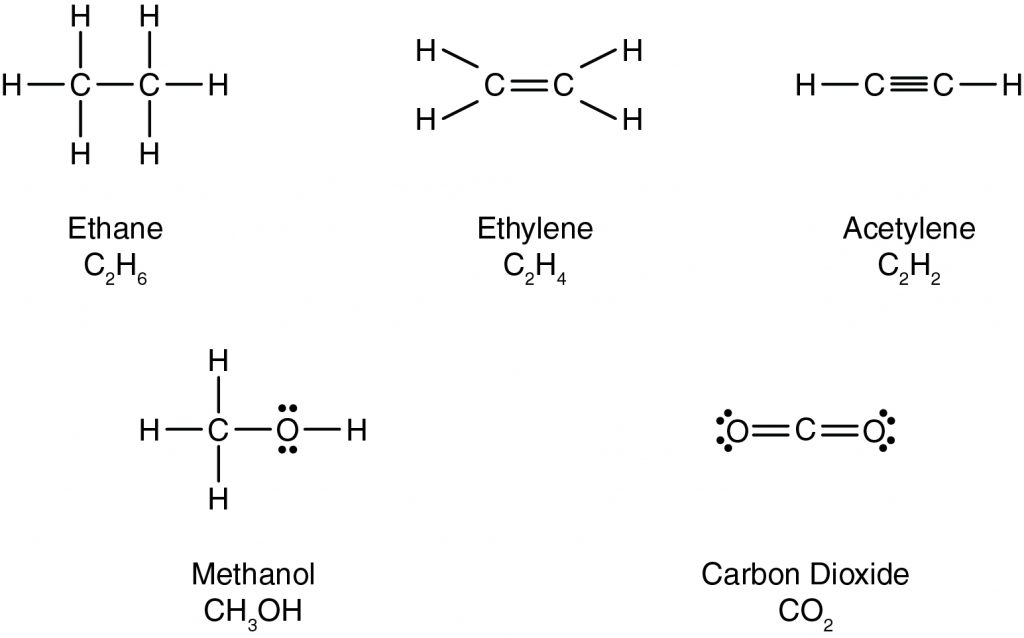 CorrectIncorrect
CorrectIncorrect -
Question 35 of 136
35. Question
In the dimensional analysis below, what is the unit of the final answer?
\(\frac{(2.25\times10^{24} atoms Pd)}{1}\times\frac{(1 mol Pd)}{(6.02\times10^{23} atoms Pd)}\times\frac{(106.42 g Pd)}{(1 mol Pd)}=398\) ?
CorrectIncorrect -
Question 36 of 136
36. Question
A reaction requires 0.446 moles of calcium to produce a desired quantity of 25.0 grams of calcium oxide (CaO). What mass of calcium is required for this reaction?
CorrectIncorrect -
Question 37 of 136
37. Question
In the dimensional analysis shown below, which unit(s) cancel out?
\(\frac{(42.5 g NH_3)}{1}\times\frac{(1 mol NH_3)}{(17.04 g NH_3 )}\times\frac{(6.02\times10^{23} molecules NH_3)}{(1 mol NH_3 )}=1.50\times10^{24} molecules NH_3\)
CorrectIncorrect -
Question 38 of 136
38. Question
A reaction requires \(8.41 \times 10^{23}\) molecules of phosphorus trichloride \((PCl_3)\). What mass of \((PCl_3)\) is needed?
CorrectIncorrect -
Question 39 of 136
39. Question
Which statement is true about 1 mole of carbon monoxide (CO)? (Refer to the Periodic Table of Elements).
CorrectIncorrect -
Question 40 of 136
40. Question
A balloon at a birthday party contains 1.05 grams of helium. How many atoms of helium are in the balloon?
CorrectIncorrect -
Question 41 of 136
41. Question
A roll of aluminum foil has a mass of 340. grams. How many moles are in the roll of aluminum?
CorrectIncorrect -
Question 42 of 136
42. Question
How many molecules are in 0.470 moles of silicon tetrachloride \((SiCl_4)\)?
CorrectIncorrect -
Question 43 of 136
43. Question
How many moles of magnesium oxide (MgO) are in a sample containing \(4.36 \times 10^{23}\) particles?
CorrectIncorrect -
Question 44 of 136
44. Question
One mole of iron is equal to _____.
CorrectIncorrect -
Question 45 of 136
45. Question
What is the unit for the final answer in the dimensional analysis shown below?
\(\frac{(48.2 g FeCl_3)}{1}\times\frac{(1 mol FeCl_3)}{(162.20 g FeCl_3 )}\times\frac{(3 mol Cl_2)}{(2 mol FeCl_3 )}\times\frac{(70.9 g Cl_2)}{(1 mol Cl_2 )}=31.6 \)?
CorrectIncorrect -
Question 46 of 136
46. Question
What is the molar mass of \(Fe(OH)_2\)?
CorrectIncorrect -
Question 47 of 136
47. Question
Ethanol \((C_2H_5OH)\) is burned as fuel in the presence of oxygen to produce carbon dioxide \((CO_2)\) and water \((H_2O)\). According to the balanced chemical reaction shown below, two molecules of ethanol require _____ molecules of oxygen to react and produce _____ molecules of carbon dioxide and _____ molecules of water.
\(C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O\)
CorrectIncorrect -
Question 48 of 136
48. Question
A reaction requires 0.35 moles of bromine \((Br_2)\). If bromine has a density of 3.1 g/mL, how many milliliters are required?
CorrectIncorrect -
Question 49 of 136
49. Question
Propane gas \((C_3H_8)\) combusts according to the equation below. How many grams of carbon dioxide \((CO_2)\) are produced from 100. grams of propane?
\(C_3H_8 + 5O_2 \rightarrow 3CO_2 + 4H_2O\)
CorrectIncorrect -
Question 50 of 136
50. Question
A can of cola contains 106 grams of sugar (\(C_{12}H_{22}O_{11}\)). Explain why it contains more or less than 1 mole of sugar.
CorrectIncorrect -
Question 51 of 136
51. Question
Aluminum and chlorine react according to the reaction below. How many molecules of chlorine \((Cl_2)\) are required to form 28 grams of aluminum chloride \((AlCl_3)\)?
\(2Al + 3Cl_2 \rightarrow 2AlCl_3\)
CorrectIncorrect -
Question 52 of 136
52. Question
Aluminum and chlorine react according to the reaction below. How many molecules of chlorine \((Cl_2)\) are required to form 28 grams of aluminum chloride \((AlCl_3)\)?
\(2Al + 3Cl_2 \rightarrow 2AlCl_3\)
CorrectIncorrect -
Question 53 of 136
53. Question
In the chemical equation below, which element(s) are unbalanced?
\(Ba(C_2H_3O_2)_2 + Na_2SO_4 \rightarrow NaC_2H_3O_2 + BaSO_4\)
CorrectIncorrect -
Question 54 of 136
54. Question
Which of the following conversion factors will be used to complete the dimensional analysis, given the balanced equation below?
\(Mg + 2HCl \rightarrow MgCl_2 + H_2\)
\(\frac{0.75 mol \hspace{0.2mm}HCl}{1}\times\frac{( ? )}{( ? )}\times\frac{24.31 g\hspace{0.2mm} Mg}{1 mol\hspace{0.2mm} Mg}\)
CorrectIncorrect -
Question 55 of 136
55. Question
If iron is exposed to oxygen, over time it will form iron (III) oxide \((Fe_2O_3)\), also known as rust. The reaction for this equation is represented by the equation below. When the equation is balanced, what are coefficients for Fe, \(O_2\), and \(Fe_2O_3\), respectively?
___\(Fe +\) ___\(O_2 \rightarrow\) ___\(Fe_2O_3\)
CorrectIncorrect -
Question 56 of 136
56. Question
Which of the following equations is balanced?
CorrectIncorrect -
Question 57 of 136
57. Question
The combustion reaction for butane (C₄H₁₀), the fuel inside lighters, is shown below. What coefficient must be added to oxygen (O₂) to balance this equation?
2C₄H₁₀ + ___ O₂ → 8CO₂ + 10H₂O
CorrectIncorrect -
Question 58 of 136
58. Question
Which unit remains after others are canceled out in the dimensional analysis shown below?
\(\frac{(12.5 g\hspace{0.2mm} NaOH}{1}\times\frac{1 mol\hspace{0.2mm} NaOH}{40.0 g\hspace{0.2mm} NaOH}\times\frac{6.02\times10^{23} particles\hspace{0.2mm} NaOH}{1 mol\hspace{0.2mm} NaOH}=1.88\times10^{23}\)particles NaOH
CorrectIncorrect -
Question 59 of 136
59. Question
What is the molar mass of \(CuSO_4\)?
CorrectIncorrect -
Question 60 of 136
60. Question
A student has attempted to balance the equation for the single-replacement reaction shown below, but it is still unbalanced. Which statement best describes how this equation is still unbalanced?
Mg + 2HCl → MgCl₂ + 2H₂
CorrectIncorrect -
Question 61 of 136
61. Question
Silver reacts with oxygen according to the balanced equation below. How many moles of silver oxide \(Ag_2O\) are produced from 9.62 moles of silver (Ag)?
\(4Ag + O_2 \rightarrow 2Ag_2O\)
CorrectIncorrect -
Question 62 of 136
62. Question
Fluorine reacts with potassium chloride (KCl) to produce potassium fluoride (KF) and chlorine gas. What mass of fluorine is required to react with 5.60 mol KCl?
\(F_2 + 2KCl \rightarrow 2KF + Cl_2\)
CorrectIncorrect -
Question 63 of 136
63. Question
A water bottle contains 500 mL of water, which has a mass of 500 grams. Which conversion factor should be used to calculate the number of moles of water in the bottle?
CorrectIncorrect -
Question 64 of 136
64. Question
The chemical equation for the synthesis of tetraphosphorus pentoxide \((P_4O_{10})\) is shown below. How many moles of oxygen \((O_2)\) are required to form 200. grams of \( (P_4O_{10})\)?
\(P4 + 5O2 \rightarrow P_4O_{10}\)
CorrectIncorrect -
Question 65 of 136
65. Question
Ammonia and oxygen react to form nitrogen dioxide and water according to the chemical equation shown below. How many grams of \(NO_2\) are produced when 2.00 moles of oxygen are consumed?
\(4NH_3 + 7O_2 \rightarrow 4NO_2 + 6H_2O\)
CorrectIncorrect -
Question 66 of 136
66. Question
Which question requires the following dimensional analysis?
\(\frac{(200.g \hspace{0.2mm}SCl_2)}{1}\times \frac{(1 mol\hspace{0.2mm} SCl_2)}{102.96 g \hspace{0.2mm}SCl_2}\times\frac{6.02\times10^{23} molecules \hspace{0.2mm}SCl_2}{(1 mol \hspace{0.2mm}SCl_2 }=1.17\times10^{24}\) molecules \(SCl_2\)
CorrectIncorrect -
Question 67 of 136
67. Question
Which equation needs to be balanced?
CorrectIncorrect -
Question 68 of 136
68. Question
In the chemical equation below, which element is unbalanced?
\(2HNO_3 + Na \rightarrow 2NaNO_3 + H_2\)
CorrectIncorrect -
Question 69 of 136
69. Question
Which of the following compounds has the lowest molar mass?
CorrectIncorrect -
Question 70 of 136
70. Question
When iron reacts with oxygen, iron (III) oxide (Fe₂O₃), also known as rust, forms according to the equation below. The iron (III) oxide has less energy than the iron and oxygen. How is reaction classified, and why?
4Fe(s) + 3O₂(g) → 2Fe₂O₃(s)
CorrectIncorrect -
Question 71 of 136
71. Question
Which of the following equations shows a decomposition reaction?
CorrectIncorrect -
Question 72 of 136
72. Question
If 70 grams of CaCl₂ are added to 100 grams of water at 10°C, what type of mixture forms, and why?
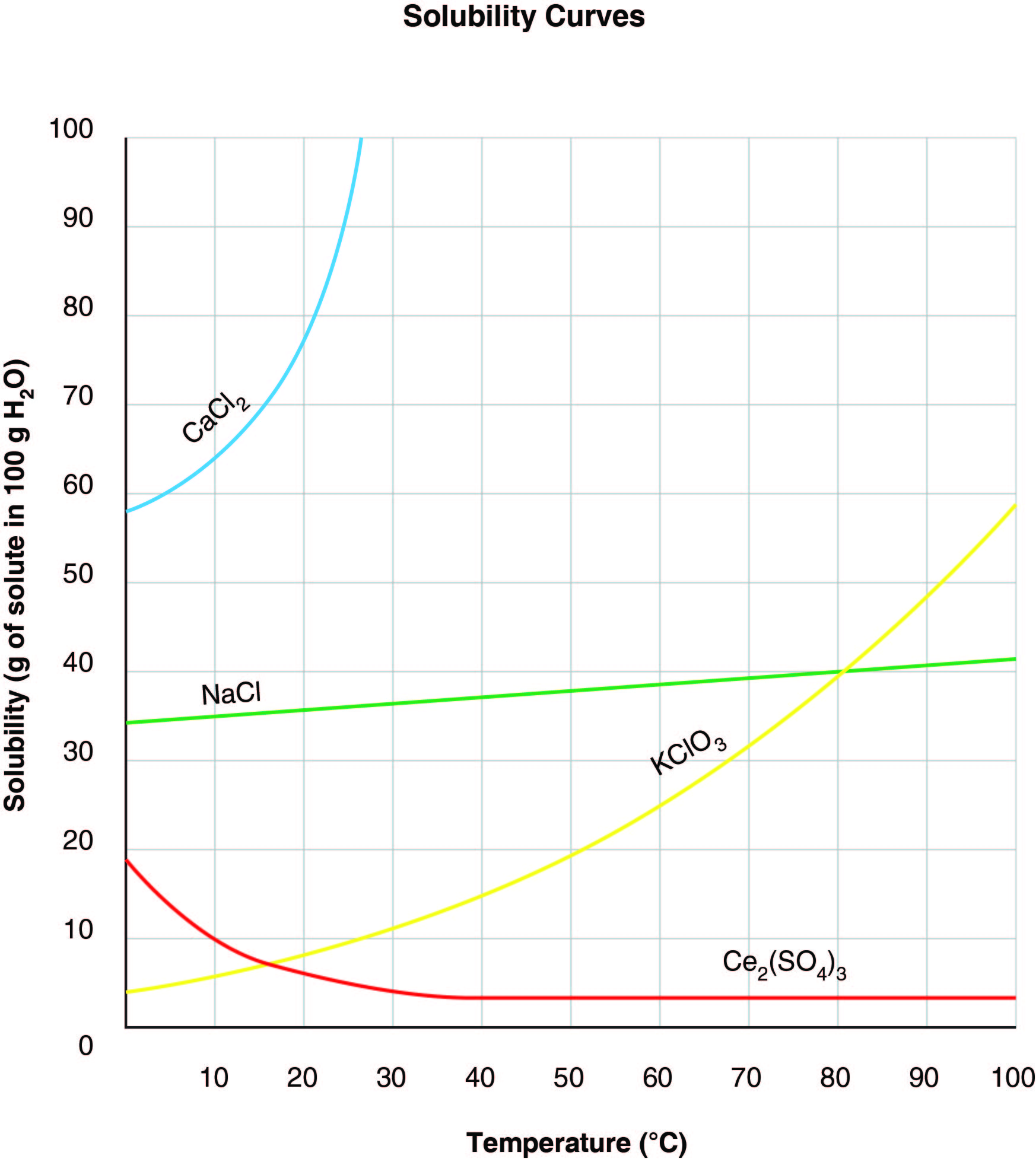 CorrectIncorrect
CorrectIncorrect -
Question 73 of 136
73. Question
The graph below shows the energy of the species involved in a chemical reaction as the reaction proceeds. How much energy is released in this reaction?
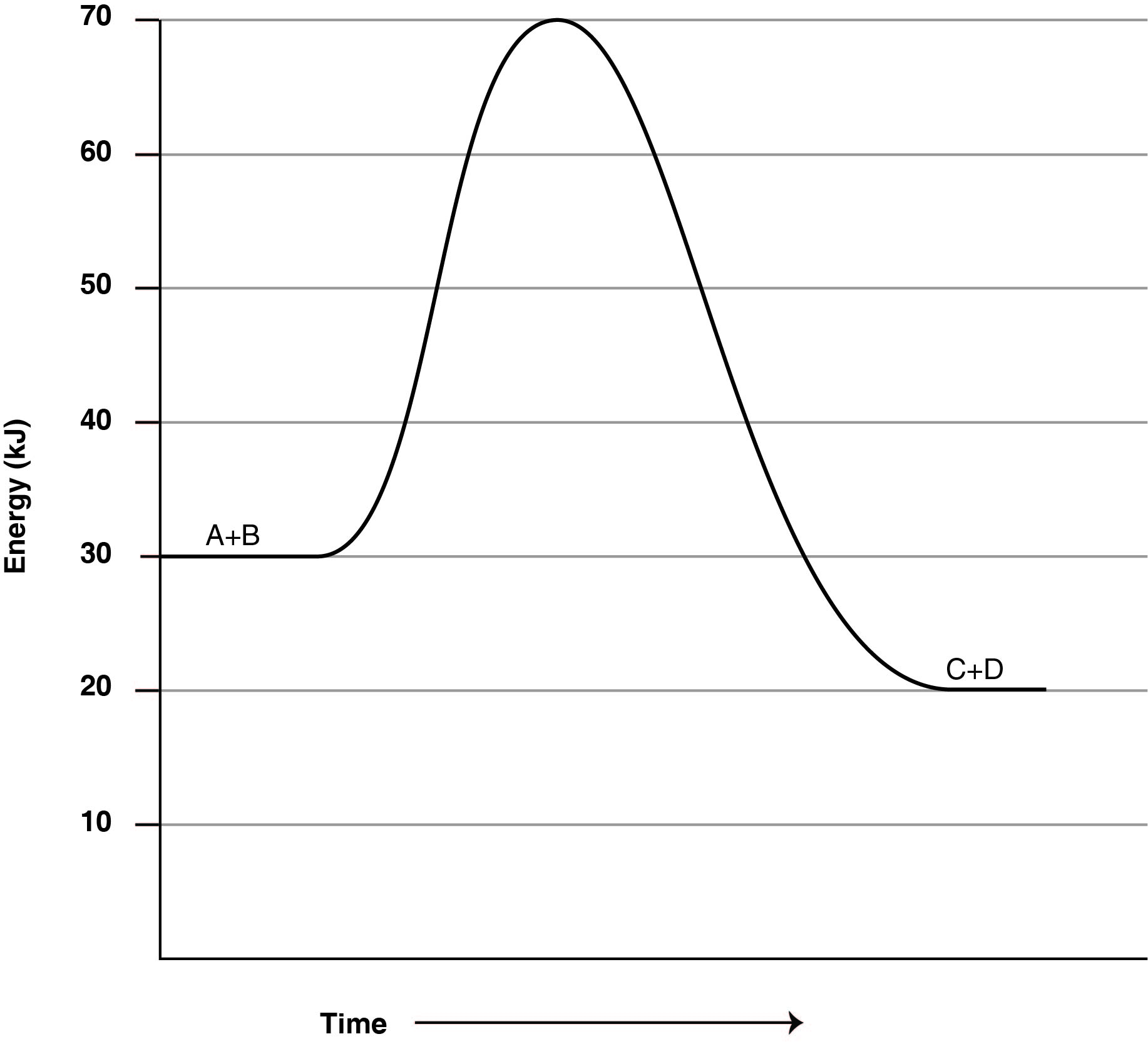 CorrectIncorrect
CorrectIncorrect -
Question 74 of 136
74. Question
What is the maximum amount of KClO₃ that can be dissolved in 50 grams of water at 30°C?
 CorrectIncorrect
CorrectIncorrect -
Question 75 of 136
75. Question
Which of the following equations shows a single-replacement reaction?
CorrectIncorrect -
Question 76 of 136
76. Question
If barium metal (Ba) and aluminum chloride (AlCl₃) react in a single-replacement reaction, what product(s) form?
CorrectIncorrect -
Question 77 of 136
77. Question
The reaction between zinc metal and hydrochloric acid (HCl) is shown below. Which product is dissolved in water at the end of the reaction?
Zn(s) + 2HCl(aq) → H₂(g) + ZnCl₂(aq)
CorrectIncorrect -
Question 78 of 136
78. Question
When an antacid tablet is placed in water, fizzing is observed. The chemical equation for the reaction that occurs is shown below. Which compound is being released in the bubbles?
C₆H₈O₇(aq) + 3NaHCO₃(aq) → 3CO₂(g) + 3H₂O(l) + NaC₆H₅O₇(aq)
CorrectIncorrect -
Question 79 of 136
79. Question
What type of reaction is described by the following equation?
2Na(s) + ZnCl₂(aq) → Zn(s) + 2NaCl(aq)
CorrectIncorrect -
Question 80 of 136
80. Question
What products are formed in all combustion reactions?
CorrectIncorrect -
Question 81 of 136
81. Question
Which compound involved in the reaction shown below is insoluble in water?
2AgNO₃(aq) + CuCl₂(aq) → 2AgCl(s) + Cu(NO₃)₂(aq)
CorrectIncorrect -
Question 82 of 136
82. Question
If 30 grams of KClO₃ is added to 100 grams of water at 0°C, some of the solute remains undissolved. What is the minimum temperature that must be attained to dissolve all the solute?
 CorrectIncorrect
CorrectIncorrect -
Question 83 of 136
83. Question
Which of the following statements regarding the solubility of Ce₂(SO₄)₃ is true?
 CorrectIncorrect
CorrectIncorrect -
Question 84 of 136
84. Question
When aqueous solutions of Pb(NO₃)₂ and KI are mixed, the reaction shown in the equation occurs, and a yellow solid appears. What is the identity of the solid?
Pb(NO₃)₂(aq)+ 2KI(aq) → PbI₂(s) + 2KNO₃(aq)
CorrectIncorrect -
Question 85 of 136
85. Question
In which reaction are both the reactants and the products heterogeneous mixtures?
CorrectIncorrect -
Question 86 of 136
86. Question
If sulfur trioxide (SO₃) reacts with water (H₂O) in a synthesis reaction, what product(s) form?
CorrectIncorrect -
Question 87 of 136
87. Question
In which reaction do the reactants form a homogeneous mixture and the products form a heterogeneous mixture?
CorrectIncorrect -
Question 88 of 136
88. Question
The equation describing the formation of ammonia (NH₃) from nitrogen and hydrogen is shown below. Which of the following statements is true?
3H₂(g) + N₂(g) → 2NH₃(g)
CorrectIncorrect -
Question 89 of 136
89. Question
What type of reaction will occur between hexane (C₆H₁₄) and oxygen (O₂)?
CorrectIncorrect -
Question 90 of 136
90. Question
The graph below shows the energy of the species involved in a chemical reaction as the reaction proceeds. Which of the following has the lowest amount of energy?
 CorrectIncorrect
CorrectIncorrect -
Question 91 of 136
91. Question
When 15 grams of Ce₂(SO₄)₃ is added to 100 grams of water at 10 ℃, some of the solute remains undissolved. How much Ce₂(SO₄)₃ remains undissolved and settles at the bottom of the beaker?
 CorrectIncorrect
CorrectIncorrect -
Question 92 of 136
92. Question
What type of reaction is described by the following equation?
2Mg(s) + O₂(g) → 2MgO(s)
CorrectIncorrect -
Question 93 of 136
93. Question
The graph below shows the energy of the species involved in a chemical reaction as the reaction proceeds. What is the activation energy required in this reaction?
 CorrectIncorrect
CorrectIncorrect -
Question 94 of 136
94. Question
Which of the following equations shows a double replacement reaction?
CorrectIncorrect -
Question 95 of 136
95. Question
Inside a plant, water has to travel up, against gravity, to reach all the leaves. What property does this illustrate?
CorrectIncorrect -
Question 96 of 136
96. Question
Why is the molecule below considered polar?
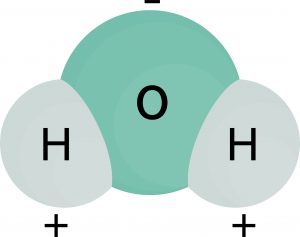 CorrectIncorrect
CorrectIncorrect -
Question 97 of 136
97. Question
A solution of silver nitrate can react with small copper beads to form silver metal and a solution of copper (II) nitrate. Which of the following changes will increase the force of collisions between reactants?
\(2AgNO_3(aq) + Cu(s) \rightarrow Cu(NO_3)_2(aq) + 2Ag(s)\)
CorrectIncorrect -
Question 98 of 136
98. Question
For the reversible reaction shown below, which of the following statements is always true of the system at equilibrium?
\(N_2 + 3H_2 \leftrightarrow 2NH_3\)
CorrectIncorrect -
Question 99 of 136
99. Question
Which of the following statements is true regarding the rates of the forward and reverse reactions for the following reversible reaction?
\(CaCO_3(s) \leftrightarrow CaO(s) + CO_2(g)\)
CorrectIncorrect -
Question 100 of 136
100. Question
The energy diagram below shows two possible pathways for a reaction: one catalyzed and one uncatalyzed. Which pathway represents a reaction that will proceed faster, and why?
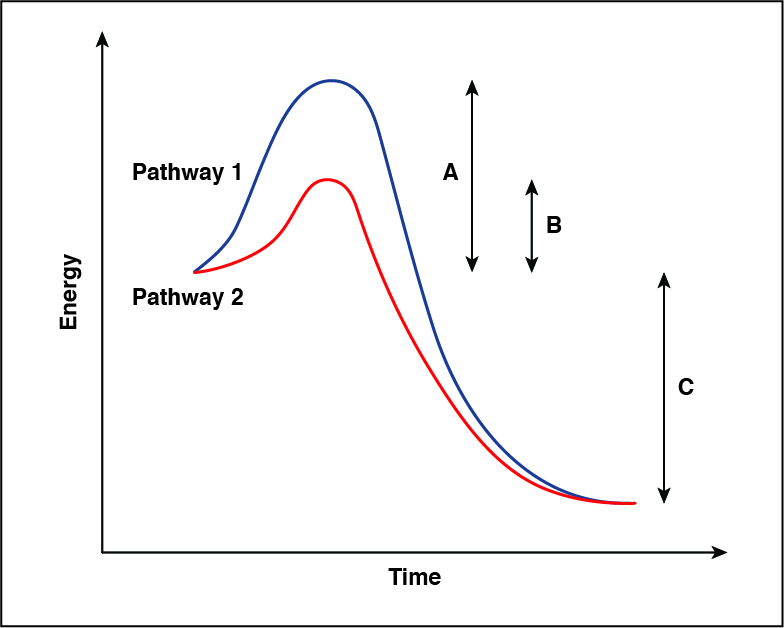 CorrectIncorrect
CorrectIncorrect -
Question 101 of 136
101. Question
Two glucose molecules (\(C_6H_{12}O_6\)) react to form a molecule of maltose (\(C_{12}H_{22}O_{11}\)) and water (\(H_2O\)). The concentrations of the reactants and products are monitored, and the data is shown below. How can the data be used to determine that this is a completion rather than a reversible reaction?
\(2C_6H_{12}O_6 \rightarrow C_{12}H_{22}O_{11} + H_2O\)
\begin{array}{|c|c|c|} \hline \textbf{Time (min)} & \textbf{Concentration of Glucose (M)} & \textbf{Concentration of Maltose (M)}\\ \hline 0 &4.48 &0\\ \hline 10 &2.44 &1.02\\ \hline 20 &1.04 &1.72\\ \hline 30 &0.32 &2.08\\ \hline 40 &0.04 &2.22\\ \hline 50 &0 &2.24\\ \hline \end{array}
CorrectIncorrect -
Question 102 of 136
102. Question
The energy diagram below shows two possible pathways for a reaction: one catalyzed and one uncatalyzed. Which of the following is true for Pathway 1, according to the energy diagram?
 CorrectIncorrect
CorrectIncorrect -
Question 103 of 136
103. Question
How does this graph show that this reaction is reversible?
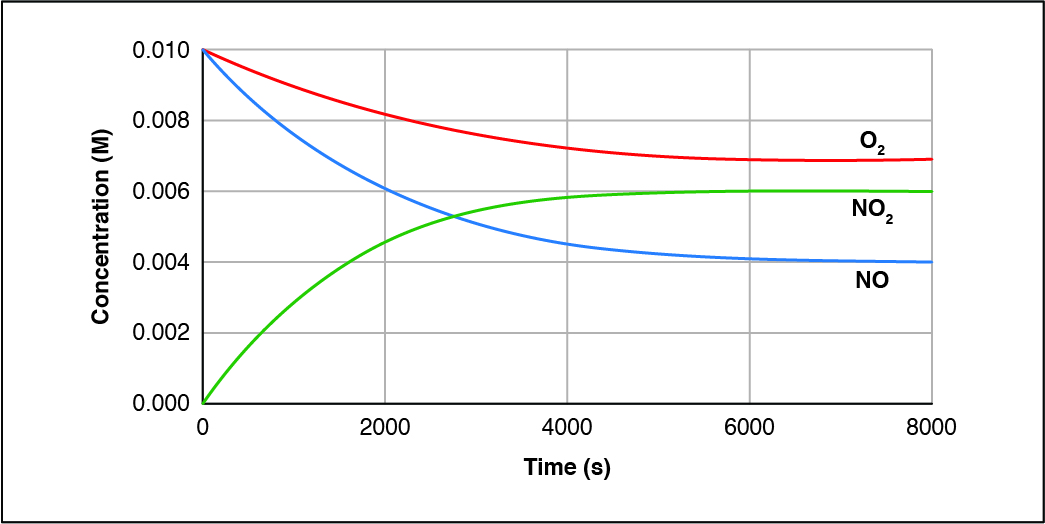 CorrectIncorrect
CorrectIncorrect -
Question 104 of 136
104. Question
The energy diagram below shows two possible pathways for a reaction: one catalyzed and one uncatalyzed. What does letter B represent?
 CorrectIncorrect
CorrectIncorrect -
Question 105 of 136
105. Question
The energy diagram below shows two possible pathways for a reaction: one catalyzed and one uncatalyzed. Which of the following is different between the catalyzed and uncatalyzed reactions?
 CorrectIncorrect
CorrectIncorrect -
Question 106 of 136
106. Question
Reaction X and Reaction Y show how fluorine (\(F_2\)) and chlorine (\(Cl_2\)) react with hydrogen (\(H_2\)). If fluorine is more reactive than chlorine, how do Reactions X and Y compare?
Reaction X: \(H_2 + F_2 \rightarrow 2HF\)
Reaction Y: \(H_2 + Cl_2 \rightarrow 2HCl\)CorrectIncorrect -
Question 107 of 136
107. Question
The graph below shows the concentrations of substances involved in a chemical reaction as the reaction progresses. Which substances are the reactants?
 CorrectIncorrect
CorrectIncorrect -
Question 108 of 136
108. Question
Which of the following statements is true regarding the role of sulfur dioxide (\(SO_2\)) in Reactions A and B?
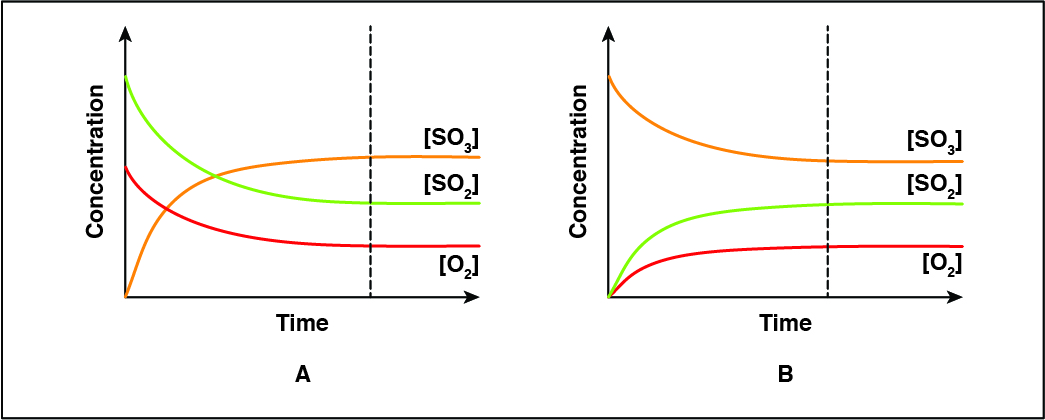 CorrectIncorrect
CorrectIncorrect -
Question 109 of 136
109. Question
An enzyme called catalase is important to many organisms because it catalyzes the decomposition of hydrogen peroxide (\(H_2O_2\)). Without this enzyme, this toxic substance would build up and damage cells. Which of the following most accurately describes how the enzyme speeds up the decomposition reaction?
CorrectIncorrect -
Question 110 of 136
110. Question
The concentrations of nitrogen, hydrogen, and ammonia were monitored in a reaction, and the data is shown below. What are the products in this reaction?
\begin{array}{|c|c|c|c|} \hline \textbf{Time (min)} &\textbf{Concentration \(of H_2\) (M)} &\textbf{Concentration of \(N_2\) (M)} & \textbf{Concentration of \(NH_3\) (M)}\\ \hline 0 &1.00 &0.25 &0.0\\ \hline 10 &0.68 &0.14 &0.22\\ \hline 20 &0.49 &0.079 &0.34\\ \hline 30 &0.38 &0.041 &0.42\\ \hline 40 &0.29 &0.025 &0.45\\ \hline 50 &0.26 &0.013 &0.47\\ \hline 60 &0.25 &0.0040 &0.48\\ \hline \end{array}
CorrectIncorrect -
Question 111 of 136
111. Question
Which of the following is true about this system at 2000 seconds?
 CorrectIncorrect
CorrectIncorrect -
Question 112 of 136
112. Question
After the reaction below reaches equilibrium, which of the following is true?
\(H_2 + I_2 \leftrightarrow 2HI\)
CorrectIncorrect -
Question 113 of 136
113. Question
Hydrogen peroxide decomposes into water and oxygen, as shown in the equation below. The rate is monitored by collecting the oxygen produced and measuring its volume over time. The data table shows the volume of oxygen collected over three minutes when two catalysts, Catalyst 1 and Catalyst 2, are used. Which catalyst results in a reaction with the lower activation energy?
\(2H_2O_2(aq) \rightarrow 2H_2O(l) + O_2(g)\)
\begin{array}{|c|ccc|} \hline \textbf{Catalyst} & &\textbf{Volume of } O_2 \textbf{ collected}&\\ \hline & 1 min & 2 min & 3 min\\ \hline 1& 1.8 mL& 2.8 mL& 3.2 mL\\ \hline 2& 1.3 mL& 1.8 mL& 2.1 mL\\ \hline \end{array}
CorrectIncorrect -
Question 114 of 136
114. Question
Which equation represents the reaction in the graph below?
 CorrectIncorrect
CorrectIncorrect -
Question 115 of 136
115. Question
The graph below shows the concentrations of substances involved in a chemical reaction as the reaction progresses. At what time is the reaction at equilibrium?
 CorrectIncorrect
CorrectIncorrect -
Question 116 of 136
116. Question
A solution of silver nitrate can react with small copper beads to form silver metal and a solution of copper (II) nitrate. Which of the following changes will increase the frequency of collisions between reactants?
\(2AgNO_3(aq) + Cu(s) \rightarrow Cu(NO_3)_2(aq) + 2Ag(s)\)
CorrectIncorrect -
Question 117 of 136
117. Question
The energy diagram below shows two possible pathways for a reaction: one catalyzed and one uncatalyzed. Which of the following is the same for the catalyzed and uncatalyzed reactions?
 CorrectIncorrect
CorrectIncorrect -
Question 118 of 136
118. Question
Which of the following statements is true regarding Reactions A and B?
 CorrectIncorrect
CorrectIncorrect -
Question 119 of 136
119. Question
Using the periodic table, determine how many protons and electrons a neutral atom of potassium has.
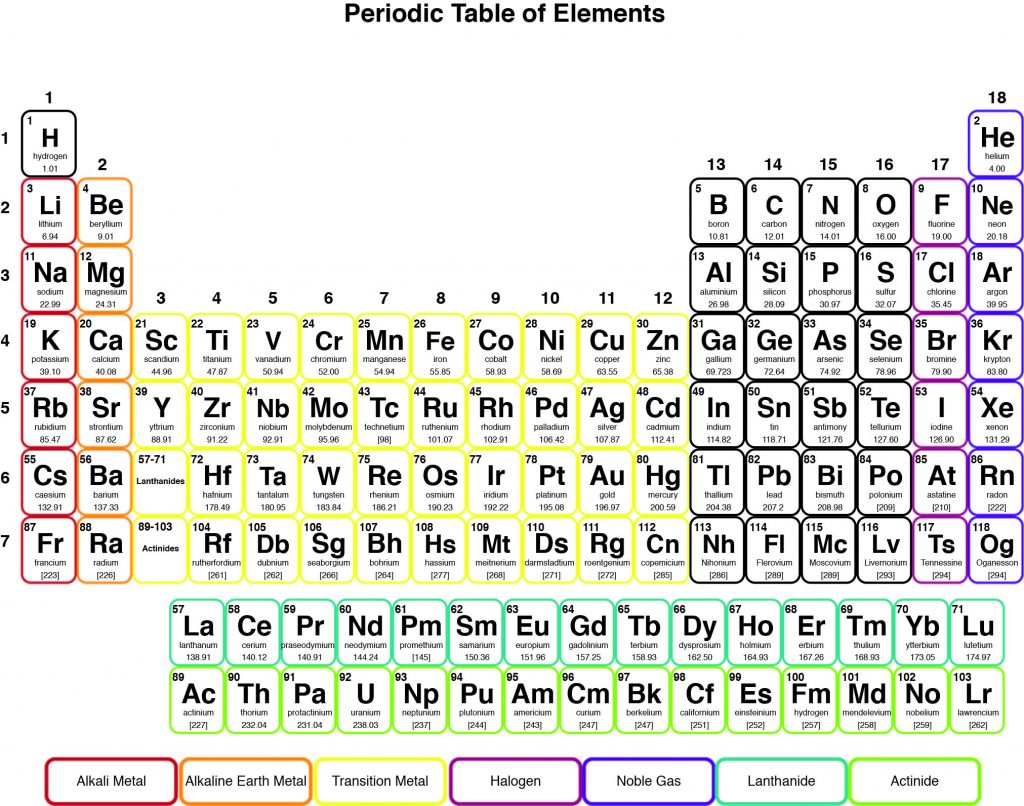 CorrectIncorrect
CorrectIncorrect -
Question 120 of 136
120. Question
Light travels at a speed of almost 3 × 10⁵ kilometers per second. How is this value written in standard notation?
CorrectIncorrect -
Question 121 of 136
121. Question
Two silicon atoms have a length of 1 nanometer, which is equal to 1 × 10⁻⁹ meter. When this value is represented in standard notation, how many zeros are between the decimal point and the nearest non-zero digit?
CorrectIncorrect -
Question 122 of 136
122. Question
The length of a year is 31,560,000 seconds. What is this value in scientific notation?
CorrectIncorrect -
Question 123 of 136
123. Question
An electron has a mass of 9.11 × 10⁻³¹ kg. When this value is represented in standard notation, how many zeros are between the decimal point and the nearest non-zero digit?
CorrectIncorrect -
Question 124 of 136
124. Question
Which of the following numbers has the smallest magnitude?
CorrectIncorrect -
Question 125 of 136
125. Question
A nanometer is equal to 1 × 10⁻⁹ meter. How is this value written in standard notation?
CorrectIncorrect -
Question 126 of 136
126. Question
The ratio of the mass of a proton to the mass of an electron is 1,840. How is this number represented in scientific notation?
CorrectIncorrect -
Question 127 of 136
127. Question
In the heating curve for iron, at which point is the substance entirely liquid?
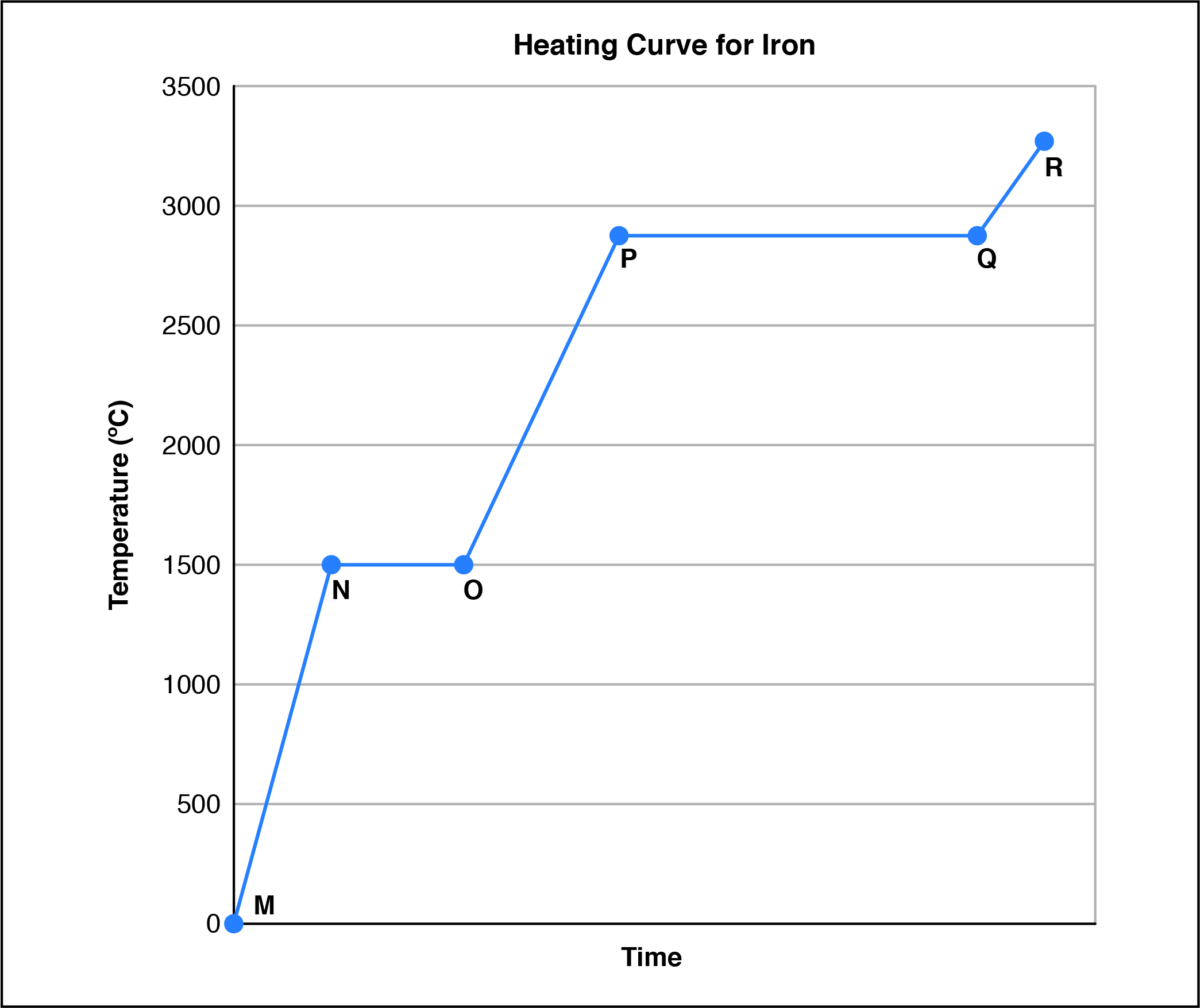 CorrectIncorrect
CorrectIncorrect -
Question 128 of 136
128. Question
Given the heating curves for fluorine, chlorine, and bromine, which halogen(s) are completely liquid at room temperature (23°C)?
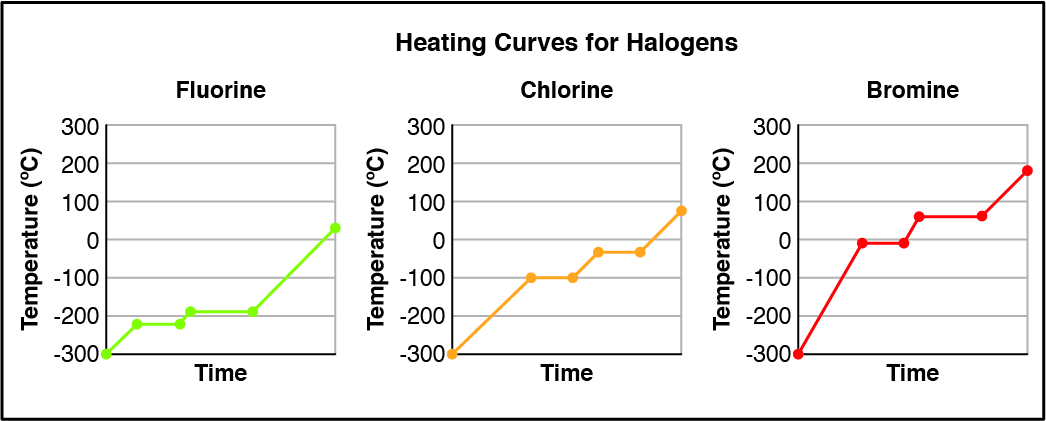 CorrectIncorrect
CorrectIncorrect -
Question 129 of 136
129. Question
Given the cooling curve below, what state(s) of matter were present at 10 minutes?
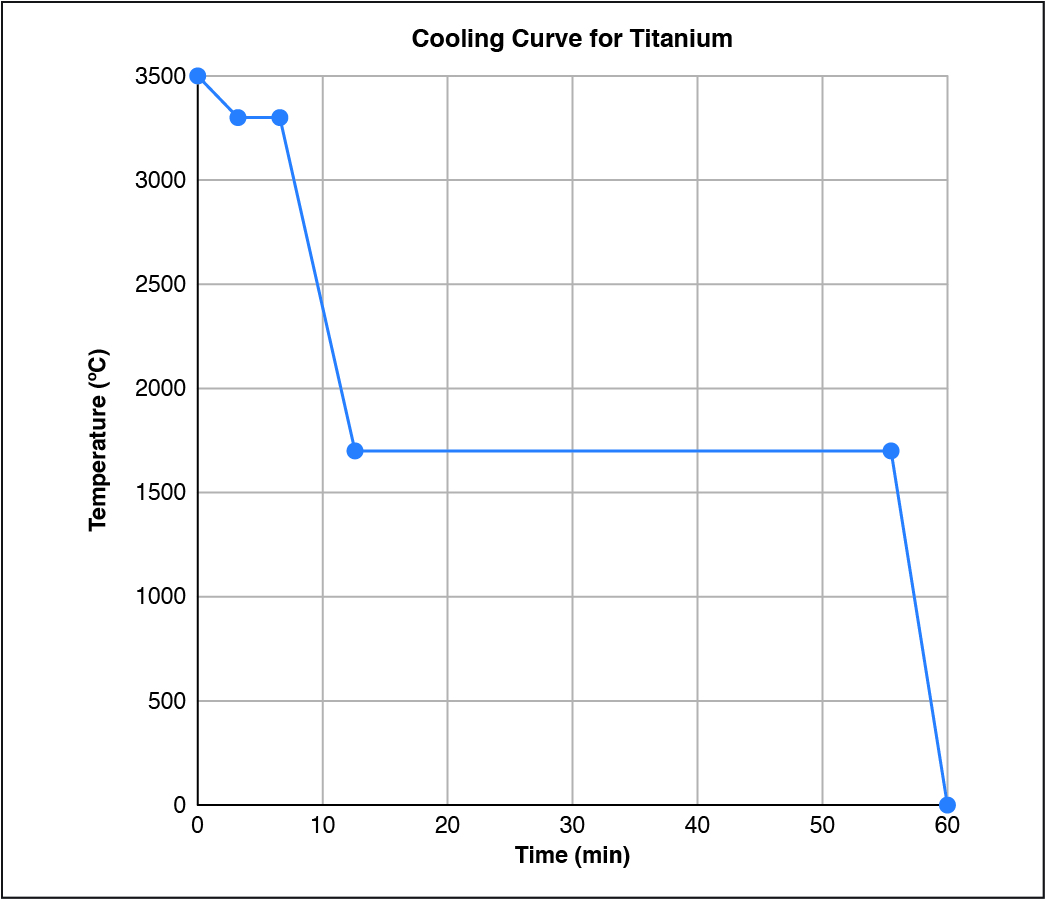 CorrectIncorrect
CorrectIncorrect -
Question 130 of 136
130. Question
According to the heating curve for iron, which process requires the most energy?
 CorrectIncorrect
CorrectIncorrect -
Question 131 of 136
131. Question
Given the cooling curve below, what is the melting point of titanium?
 CorrectIncorrect
CorrectIncorrect -
Question 132 of 136
132. Question
Given the heating curves for fluorine, chlorine, and bromine, which halogen boils at the lowest temperature?
 CorrectIncorrect
CorrectIncorrect -
Question 133 of 136
133. Question
At which temperature are all three halogens in the same state of matter?
 CorrectIncorrect
CorrectIncorrect -
Question 134 of 136
134. Question
Compare the boiling points of the alcohols in the table below. Molecules of these alcohols are similar, but they have different numbers of carbon atoms in a chain and, therefore, different masses. Based on the boiling point data in the table, what conclusion can be drawn about the relationship between the number of carbon atoms in a molecule and the interaction of the molecules?
 CorrectIncorrect
CorrectIncorrect -
Question 135 of 136
135. Question
Given the cooling curve below, what state(s) of matter were present between 20 and 30 minutes?
 CorrectIncorrect
CorrectIncorrect -
Question 136 of 136
136. Question
Given the heating curve of iron, what are the approximate melting and boiling points?
 CorrectIncorrect
CorrectIncorrect
